[INQ. NO. 2604E07] Innerwave Inc. is a software technology company that has grown through extensive experience in providing IT services both for public institutions and major corporations. Through continuous research and development, the company has expanded into the digital healthcare sector ― focusing on AI-based diagnostic support, treatment, and monitoring solutions, as well as AI healthcare platforms. Leveraging its expertise, Innerwave has concentrated on digital medical device development and related services.
Recently, Innerwave received regulatory approval from the Ministry of Food and Drug Safety (MFDS) in Korea for its digital therapeutic device for nicotine-use disorders, under the brand name of Dr. JinNicojini. This product is a mobile app–based digital therapy prescribed to patients after being diagnosed by psychiatric or family medicine specialists. It is structured as an eight-week program grounded in Cognitive Behavioral Therapy (CBT), combining coping strategies for nicotine cravings with motivational-enhancement training to support patient self-management in daily life.
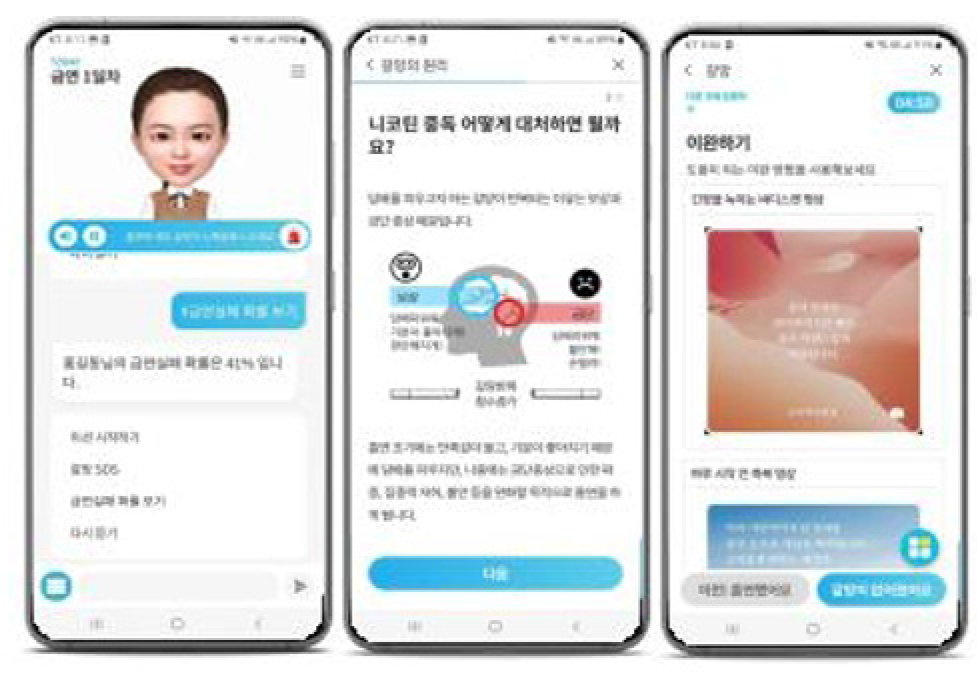
Dr. JinNicojini is a product developed to apply cutting-edge technology through innovative medical device approval ― and it has secured content differentiation based on personalized features. The app delivers personalized content through a stepwise eight-week treatment protocol designed to facilitate behavioral change. By collecting and analyzing digital patient data, it supports adherence monitoring and sustained engagement throughout the treatment process. It is significant in that it is a digital therapeutic device that has obtained regulatory approval in Korea, with the goal of improving people’s nicotine-use disorders.
Building on its experience in developing disease-management applications and patient-management platforms for high-level general hospitals, Innerwave plans to expand its domestic clinical references following commercialization. By promoting the adoption of its innovative medical technology in healthcare institutions, the company aims to accumulate real-world clinical cases and strengthen synergies with its broader AI healthcare platform business.
For its overseas expansion strategy, Innerwave has set the German Digital Health Application (DiGA) market as its primary target. To this end, it is preparing to obtain CE MDR certification, as it believes Germany is a market where the regulatory integration of digital therapeutic devices can occur relatively quickly compared to other countries. After securing sufficient domestic references, we plan to pursue a phased expansion into the European and Southeast Asian markets.
Innerwave plans to expand its portfolio of disease-specific digital therapeutic devices based on its digital medical-device development service capabilities and AI-based platform technology.
Starting with Dr. JinNicojini Genie, the company intends to advance its technological competitiveness and business model in the field of digital therapy, and accelerate its entry into the global digital healthcare market.
Innerwave has obtained regulatory approval for software medical devices, achieving development milestones in digital medical devices, mobile health, and AI disease prediction systems—backed by AI-based development infrastructure and clinical data from medical institutions. “We are committed to driving global digital biohealth innovation through our comprehensive one-stop integrated service utilizing medical platforms, AI, and Big Data Cloud technologies,” remarked the company’s CEO.

korean-electronics.com | Blog Magazine of korean electronics, brands and Goods




Leave a comment