[INQ. NO. 2212E23] RF Medical, as a medical device manufacturer, has been dedicated to providing equipment for radiofrequency ablation treatments since 2003.
RF Medical’s extensive product range includes RFA systems for tumor ablation (such as liver cancer, and thyroid nodule ablation), plus varicose vein ablation systems.
RF Medical’s sophisticated technology has allowed it to spread its business to more than 60 countries around the world, and its proven quality has earned it a strong reputation in the market. RF Medical strives to create a healthier world through continuous investment in research and development.
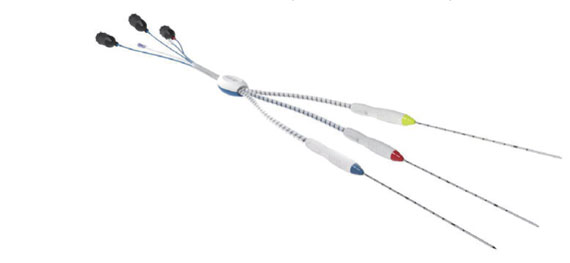
RF ablation systems consist of a radiofrequency generator and various types of radiofrequency electrodes
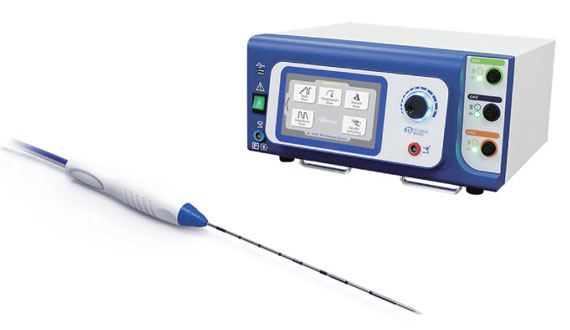
RF Medical’s RF (radiofrequency) ablation system consists of a radiofrequency generator, a cooling pump, and various types of radiofrequency electrodes. The radiofrequency ablation system is used worldwide to treat different types of soft tissue.
The use of the radiofrequency ablation system for treatment in soft tissue ablation ranges from liver tumors (HCC), to renal cancer (RCC), lung cancer, thyroid tumors, and uterine myoma, etc., plus varicose vein treatments. The RF ablation system provides patients an opportunity to get treated in a safe and minimally invasive way.
RF Medical’s RF generator M-3004 and V-1000 are CE-approved & U.S. FDA 510 (k) cleared. These generators are compatible with various types of RF electrodes that are manufactured and supplied by RF Medical. It has a user-friendly touch screen interface and can also be controlled by a foot switch, if needed.
The biggest advantage of RF’s generators is that they come with pre-loaded algorithms, such as the controlled impedance mode, temperature-controlled mode, manual mode, etc. These pre-loaded algorithms help physicians to use its RF ablation system for various kinds of indications.
RF Medical’s electrodes that can be used with M-3004 and V-1000 generators are also CE approved & U.S. FDA 510(k) cleared.
RF Medical exports its products to more than 50 countries, and its overseas sales account for 80 percent of its entire sales.
RF Medical plans to expand its portfolio of minimally invasive medical devices next year, using microwave and cryo-ablation techniques to offer a comprehensive minimally invasive solution.
korean-electronics.com | Blog Magazine of korean electronics, brands and Goods




 Medical Device for Vascular Treatment
Medical Device for Vascular Treatment Dual Wavelength Long Pulse Laser
Dual Wavelength Long Pulse Laser Non-invasive Medical Device
Non-invasive Medical Device A spokesperson for Lutronic explains, “We continue to take great leaps forward toward becoming a total healthcare provider starting at the center of new trends in the post-pandemic beauty ecosystem.” He added that “Lutronic will be establishing a stronger brand power globally by showcasing innovative products aimed at addressing the skin concerns of medical practitioners and patients around the world.”
A spokesperson for Lutronic explains, “We continue to take great leaps forward toward becoming a total healthcare provider starting at the center of new trends in the post-pandemic beauty ecosystem.” He added that “Lutronic will be establishing a stronger brand power globally by showcasing innovative products aimed at addressing the skin concerns of medical practitioners and patients around the world.”



 How to Maximize Treatment Effect of POICOCARE450
How to Maximize Treatment Effect of POICOCARE450


