[INQ. NO. 2110E20] GENO is a medical device manufacturer, with its HQ in Seoul, Korea and a branch in Tianjin, China. GENO has built distribution networks mainly focusing on overseas exports after launching a number of dental material brands that are well-received by foreign nations as they are compatible with other commercialized systems.
GENO has obtained CE and CFDA quality certifications in recognition of its outstanding technical prowess. Thanks to continuous investments made into R&D, GENO is producing not only premium implants, but also surgical suture materials and PDO lifting threads, providing customized products for overseas customers and improving services so as to distinguish itself from other rivals and create profitable structures.
GENO has designed the order of all processes including product planning, designing, production preparation and others through ‘the medical device production line automation.’ By automating production control, management, operation, and others, the company could improve quality stability.
And through vision inspection systems and PLC facilities, the company could develop ICT-based ‘industrial automatic equipment for all processes’ for the maximization of the production efficiency of medical devices, it started to take off as a device developer.
Medical material-producing devices that have been so far developed include ‘a surgical suture automation production device and a lifting thread automation production device.’ GENO has turned manual and partially automated production processes into a ‘complete automation production system.’
The CEO of GENO welcomes the keen attention from those involved in the medical device manufacturing sector for the direction the company is going to take, saying that the company is planning to provide a consignment production solution through standardized technologies.
korean-electronics.com | Blog Magazine of korean electronics, brands and Goods



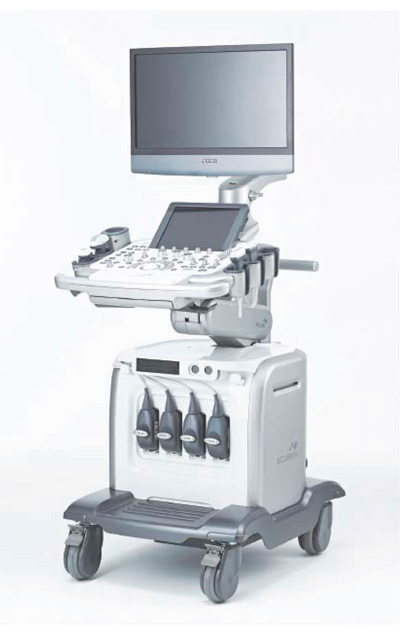 A new product, E-CUBE Platinum is a premium system that provides high quality imaging. It is embedded with software that can be applied to various body parts including the abdomen, musculoskeletal system, breast, heart, blood vessels, and uterus.
A new product, E-CUBE Platinum is a premium system that provides high quality imaging. It is embedded with software that can be applied to various body parts including the abdomen, musculoskeletal system, breast, heart, blood vessels, and uterus.
 How to Maximize Treatment Effect of POICOCARE450
How to Maximize Treatment Effect of POICOCARE450 that global leaders such as GE Healthcare and Siemens are using its grid parts.
that global leaders such as GE Healthcare and Siemens are using its grid parts.


