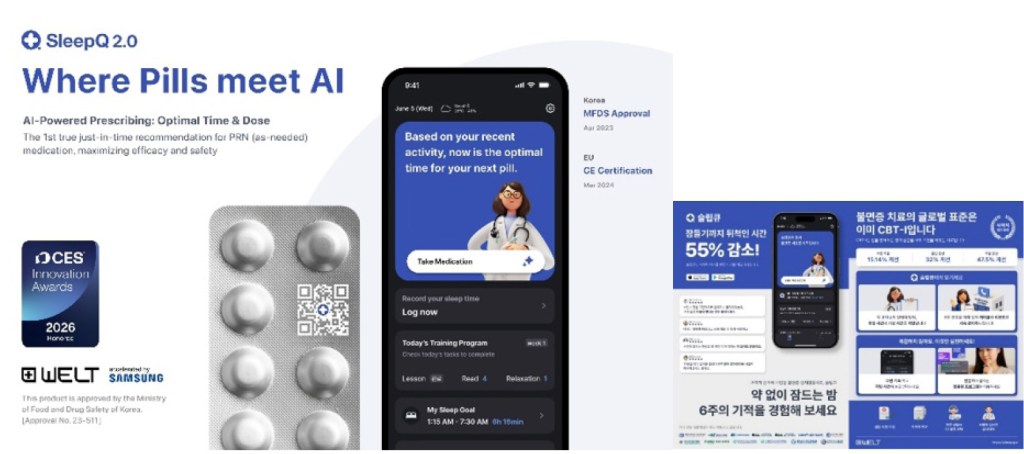
[INQ. NO. 2605E05] JSKBIOMED Inc. is a biomedical company specializing in laser-based, needle-free injection technology, developing next-generation drug delivery solutions that address the limitations of conventional injection methods ― including pain, infection risk, and medical-waste generation.
Based on proprietary technology that precisely controls the skin barrier using laser energy, the company has established a platform capable of effectively delivering drugs, vaccines, and functional substances, with scalability across medical, aesthetic, and healthcare fields.

Following the stabilization of its core technology in 2025, JSKBIOMED has begun full-scale expansion into overseas markets, entering major countries including Japan, Poland, and Sweden. In Korea, the technology is currently being applied in approximately 300 hospitals and clinics, thereby, demonstrating both technical stability and clinical applicability.
JSKBIOMED recently introduced its laser-based, needle-free injection technology and new product lineup ― aiming to expand strategic partnerships with overseas distributors, medical device companies, and pharmaceutical and biotech firms. The company has been seeking to release collaboration models tailored to country-specific distribution structures and market characteristics while building long-term global networks.
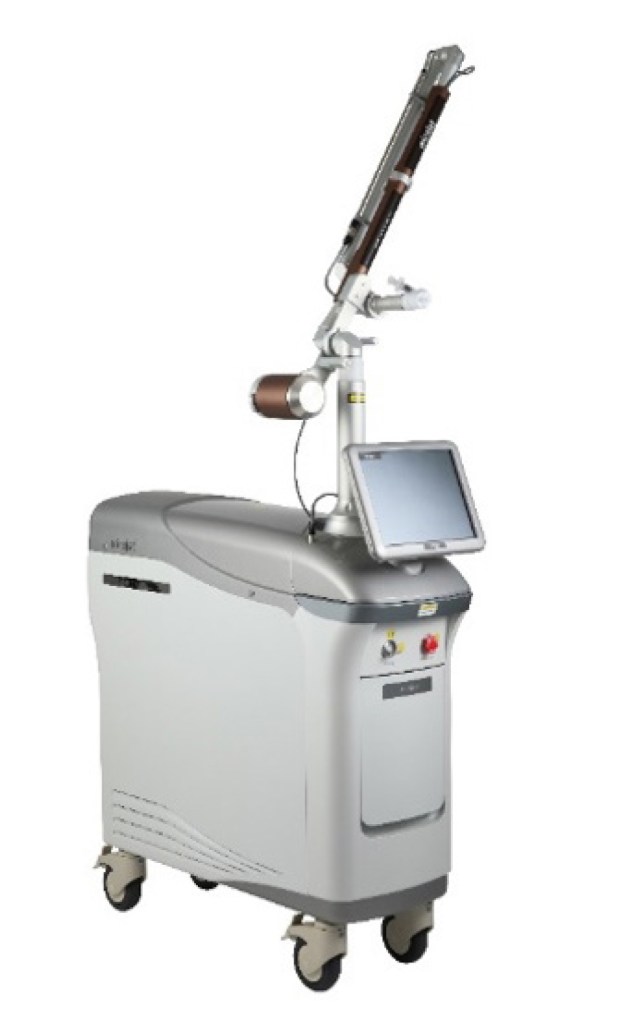
JSKBIOMED’s flagship medical product, MiraJet, utilizes laser-based needle-free injection technology to deliver drugs and active ingredients effectively while minimizing skin damage. It is designed to reduce pain and infection risk while lowering dependency on practitioner skill, thereby enabling a more consistent treatment environment.
JSKBIOMED also recently introduced its aesthetic equipment Jetical Pro 2940, which presents a solution optimized for professional treatment settings. Based on precise laser- control technology, it supports stable energy delivery and enhanced treatment control, and is designed to improve skin condition and increase the efficiency of active ingredient delivery. It can be applied to various skin types and treatment purposes, expanding its usability in professional clinics.

In addition, JSKBIOMED is expanding into the homecare market with the Jetical Mini Pro, a consumer device that integrates the core technology of its medical and professional equipment. Designed with enhanced usability and safety, it enables personalized skincare management, forming an integrated laser-based needle-free injection solution ecosystem across medical, professional, and home-use categories.
JSKBIOMED has identified the United States, Japan, China, France, and Germany as key target markets. These countries represent mature medical and aesthetic device markets with continuously increasing demand for high-performance and high-value aesthetic technologies. The company plans to pursue phased market expansion through collaboration with local buyers, distributors, and global beauty and biotech companies.
In the first half of 2026, JSKBIOMED will be participating in major international exhibitions such as AMWC Monaco and ASLMS in order to strengthen global brand recognition, while promoting its technology and expanding partnerships.
At the same time, JSKBIOMED will pursue country-specific regulatory approvals and certifications in stages and accelerate its growth as a global laser-based needle-free injection platform company through technology licensing, joint development, and diversified collaboration models.
korean-electronics.com | Blog Magazine of korean electronics, brands and Goods