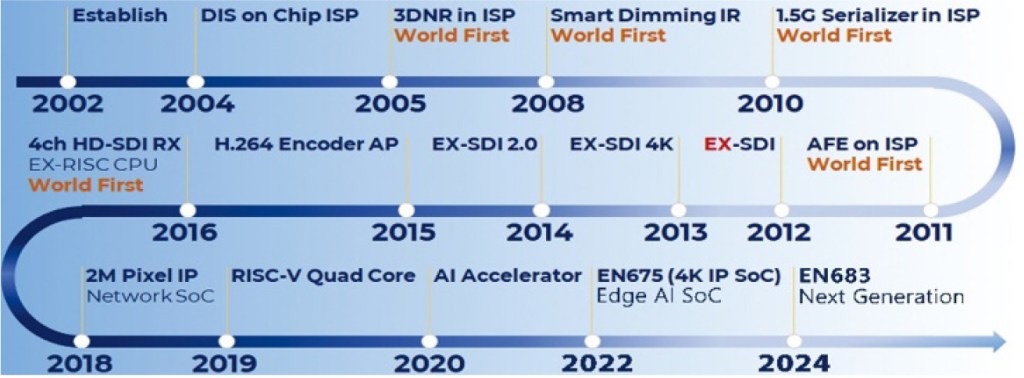
[INQ. NO. 2403E05] Founded in 2017, ICTK is a security solutions company specializing in Physical Unclonable Functions (PUF) technology. Since the company began mass-producing PUF chips shortly after its establishment, it has been building various security platforms and supplying leading domestic and foreign companies such as Doosan Robotics.
ICTK is the only company in Korea that has met all the ‘RoT’ (Root of Trust) requirements of the Internet of Things (IoT) Security Division of the Global Semiconductor Association (GSA), and our unique technology, ‘VIA PUF,’ is now officially registered with GSA.
In particular, in December 2020, PUF technology was officially established as an international standard (ISO/IEC 20897), and ICTK is looking forward to the accompanying growth of the related domestic industry. In July 2021, it was also certified as a KCMVP (Korea Cryptographic Module Validation Program) by the nation’s National Intelligence Service.
Security chip based on RoT function
The G3 is a security chip based on hardware Root of Trust (RoT) function powered by VIA PUF —ICTK’s proprietary technology, that provides security functions such as device authentication, anti-counterfeiting, firmware protection, and secure storage.
ICTK’s VIA PUF technology takes IoT supply chain security to the next level by establishing the RoT function supported by the unclonable VIA PUF technology, protecting sensitive data even from the most advanced hacking attacks. The G3 chip helps extend the life of IoT devices. It meets all ISO standards and has high competence in 13 detailed core technologies. ICTK has successfully mass-produced the world’s first PUF security chip.
ICTK solved the problem of “writing a unique key from outside” in existing memory-based security chips. It solved the homeostasis problem of existing active element-based PUF security chips. The stability and homeostasis of the company’s passive PUF chip and market validation through mass production are possible.
“We truly wish to do our best to provide a safer foundation for smart life by providing powerful security solutions based on hardware (HW), from security chips to services. Thank you!” the company’s CEO commented.
korean-electronics.com | Blog Magazine of korean electronics, brands and Goods