[INQ. NO. 2603E12] TNH Co., Ltd. is a healthcare IT company that has focused on addressing the structural challenges in healthcare. Since its founding in 2008, TNH has focused on developing specialized hospital software and has consistently provided solutions tailored to the hospital operating environment, focusing on cloud-based EMR and CRM.
TNH’s unique strength lies in expanding its solutions beyond simple electronic chart provision to encompass the entire hospital workflow, encompassing reservations, treatment, consultations, payments, insurance claims, and post-mortem care, all within a single system.

Its VegasCRM is a cloud-based EMR/CRM integrated solution for dermatology, aesthetics, and plastic surgery hospitals and clinics. It is designed to integrate not only medical record management but also reservations, customer management, and insurance claims into a single system.
Real-time status boards, treatment room status boards, and a smart reservation system allow for a comprehensive overview of hospital operations. It also offers customer management through point payment and deduction functions, and pick-up desk-based treatment distribution and statistics.
The reception and consultation function using tablet charts is also a factor that increases field usability.
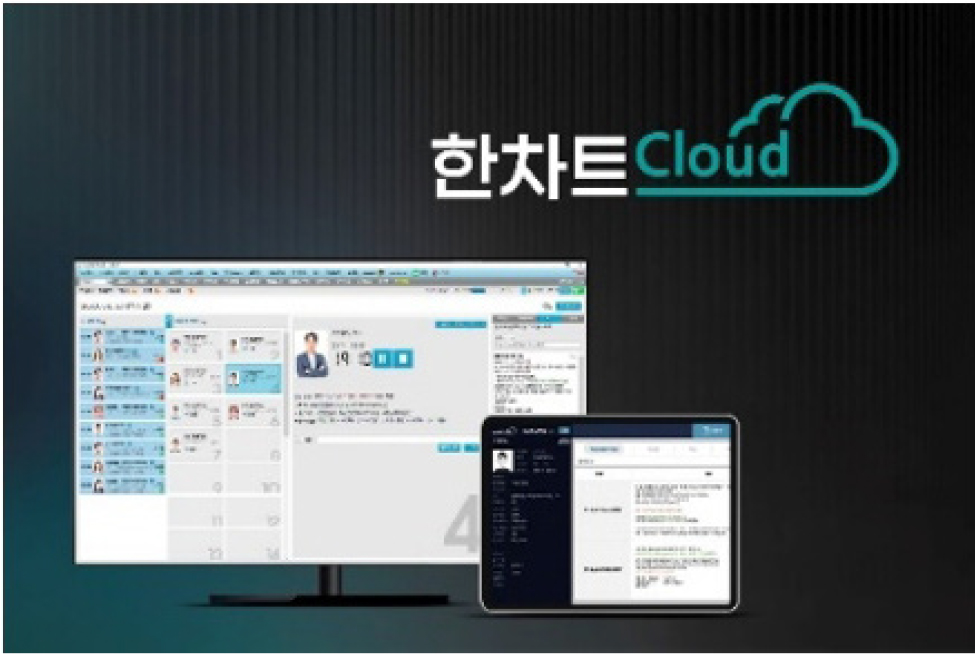
TNH’s HanChart Cloud is a cloud-based electronic chart solution designed specifically for the environment of Oriental medicine clinics. It supports stable hospital operations without space or equipment constraints. It also features enhanced features specific to Korean Oriental medicine clinics, such as beauty treatment image storage, CRM functionality, kiosk (HIOSK) integration for registration, and certificate issuance.

Furthermore, TNH plans to gradually introduce a solution with enhanced field usability and mobile scalability via the tablet-based medical app Mediclo. Through these initiatives, TNH aims to more organically connect EMR and CRM-based hospital operating environments and continue its role as a technology partner that reduces actual workloads in the medical field.
Currently, TNH’s solutions are used in over 3,000 hospitals and clinics nationwide, with over 15,000 medical professionals and hospital staff utilizing them in real time on average daily. TNH continues its steady growth, with revenue of KRW 5 billion in 2022, KRW 6.2 billion in 2023, KRW 8.1 billion in 2024, and KRW 9.2 billion in 2025. This success is attributed to long-term customer retention and solution reliability.
korean-electronics.com | Blog Magazine of korean electronics, brands and Goods