[INQ. NO. 2602E07] NTL Healthcare Co., Ltd, a medical device company specializing in the early diagnosis of female cancers, is expanding its business in the global market through diagnostic solutions that combine medical technology and artificial intelligence (AI).
Since its first foray into the field of gynecological cancer diagnosis in Korea in 1996 with the introduction of ‘Cervicography,’ an analog-based cervical cancer screening system, NTL Healthcare has continuously advanced its remote screening system and AI-based diagnostic technology.
NTL Healthcare has focused on improving the accuracy and accessibility of cervical cancer screening, focusing on developing technologies that can provide the same quality of diagnosis even in settings with inadequate existing medical infrastructure.
Based upon this accumulated technological expertise, NTL Healthcare is currently building a female disease diagnostic platform centered on its AI-based cervical cancer screening solution, ‘CerviCARE® AI.’
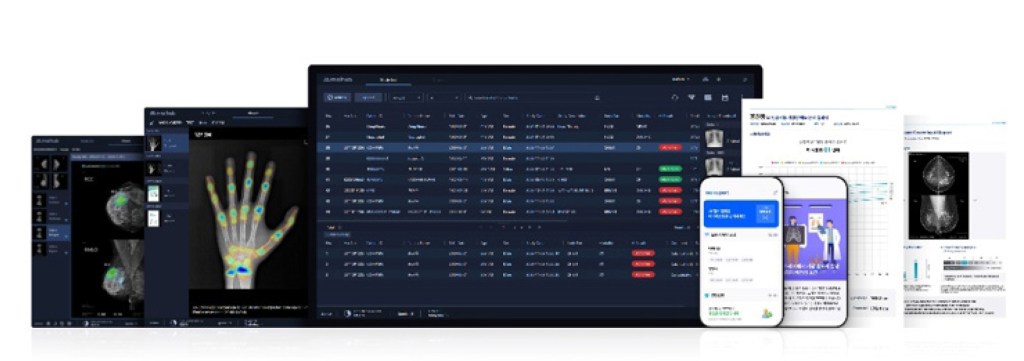
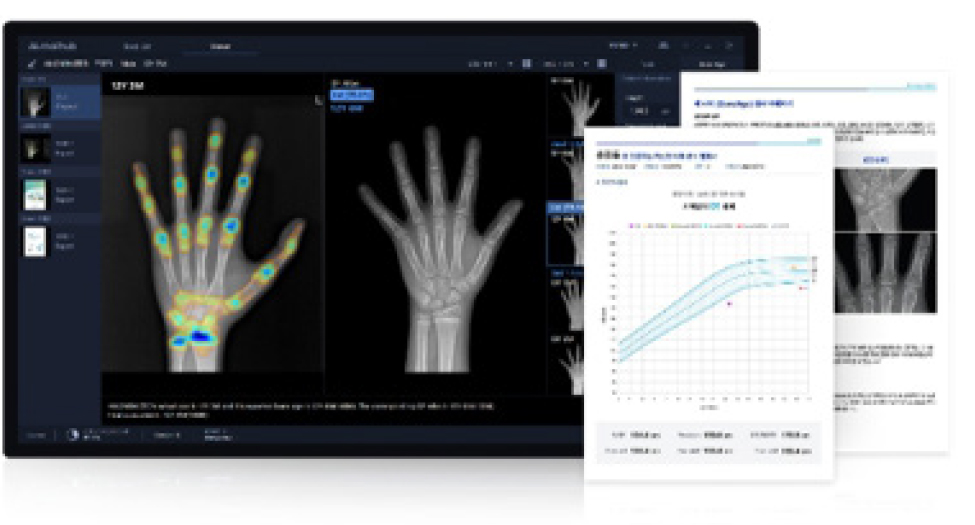
CerviCARE® AI is medical software that uses deep-learning-based AI to analyze cervical lesion images, complementing the subjective judgment of the diagnostician and providing standardized analysis results.
CerviCARE® AI is designed to capture cervical lesions externally, and then use AI to detect and analyze them to classify their condition, thereby assisting in determining subsequent treatment options. This reduces variation across testing environments and readers, thereby contributing to increased diagnostic efficiency.
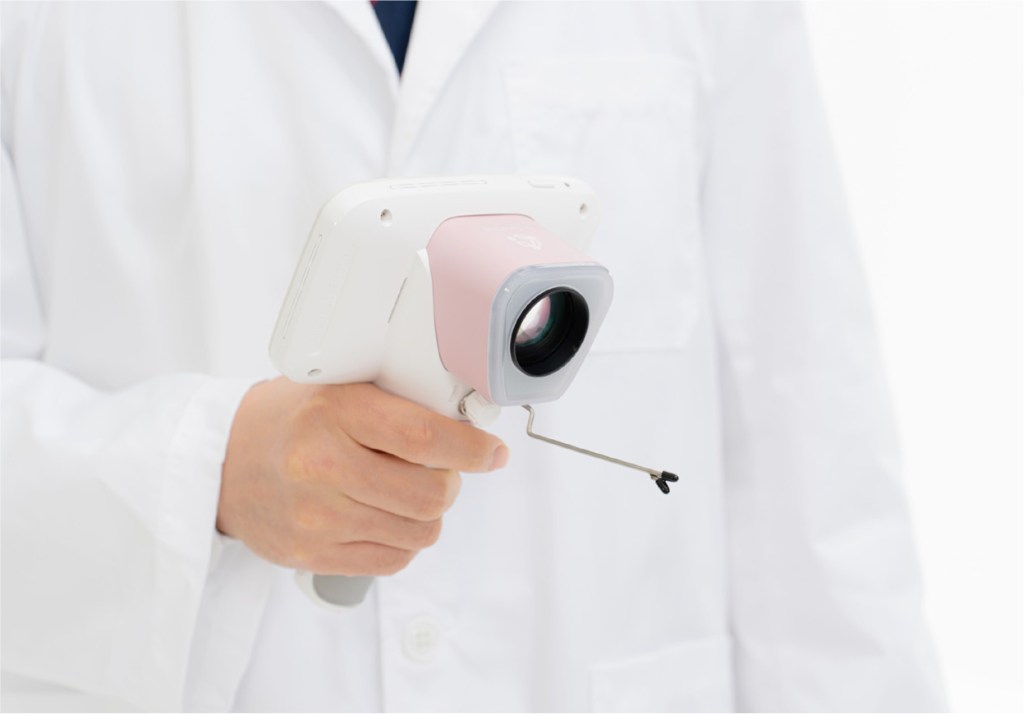
Furthermore, the solution is designed to integrate with remote diagnostic systems to support rapid decisions, even when a second opinion from an expert is required. It is also meaningful in terms of improving medical accessibility and expanding the scope of women’s health care, as it can also be utilized in environments where traditional medical facilities are lacking.
NTL Healthcare currently exports its products to approximately 20 countries and has been gradually expanding its global presence, focusing on Asian countries such as Thailand, Mongolia, and Vietnam. Building on this momentum, it is also pursuing expansion into low- and middle-income countries (LMICs) and the European market.
Through its participation in this KIMES exhibition, NTL Healthcare is seeking to discover new business partners and expand global partnerships through direct consultations with domestic and international medical device buyers, distributors, and hospital officials. Through on-site demonstrations and consultations, NTL Healthcare plans to directly demonstrate the product’s functionality and usability, incorporating market needs and the latest trends into future product development and marketing strategies.

NTL Healthcare is prioritizing Japan, Indonesia, and Australia as its key markets, and is expanding collaboration opportunities in countries with a high level of acceptance of new medical technology.
In particular, CerviCARE® AI is a solution that can be utilized both in regions with limited medical infrastructure and advanced medical environments. Its strength is evaluated as its ability to simultaneously ensure standardization and speed in the process of diagnosis.
NTL Healthcare plans to continue participating in various domestic and international medical device exhibitions and academic events, including MEDICA 2026. Through these activities, NTL Healthcare aims to strengthen its networking with global medical experts and the industry persons concerned, and further promote the technology’s clinical value and potential applications.
Based on these activities, NTL Healthcare plans to continue expanding its services to provide more efficient and standardized medical services in the field of female cancer diagnosis.

korean-electronics.com | Blog Magazine of korean electronics, brands and Goods