 [INQ. NO. 2002E14] Founded in 1993, MEDIANA’s Co., Ltd. is a specialized manufacturer of medical devices, and it exports ODM as well as its own branded products to clients all over the world.
[INQ. NO. 2002E14] Founded in 1993, MEDIANA’s Co., Ltd. is a specialized manufacturer of medical devices, and it exports ODM as well as its own branded products to clients all over the world.
MEDIANA’s advanced body composition analyzer i30 provides an accurate analysis of body composition and body water distribution through multi-frequency measurement.
It measures the resistance value of cell membranes through an electric current impedance analyzing system. It accurately measures segmental body fat and muscle mass. It has a high correlation with the so-called gold standard devices such as DEXA, CT and isotope dilution analysis system. Top medical professionals have proven the clinical reliability of i30.

The on-screen scale imaging system enables more familiar, traditional analysis of body composition.
It focuses on more personalized user management based on diverse input interface, wireless data communication and DB management on its website. The ergonomically designed, rotatable hand electrodes allow the user to take a test with ease.
Mediana aims to become a truly global market leader in the medical equipment industry through continuous research and development investments, by developing new products such as monitor/ defibrillators, AEDs and patient monitors that form a major product line today.
korean-electronics.com | Blog Magazine of korean electronics, brands and Goods
 Moreover, PICOCARE received the U.S. FDA approval for the first time as a product manufactured by an Asian company and it boasts the largest market share domestically as one of the picosecond laser devices.
Moreover, PICOCARE received the U.S. FDA approval for the first time as a product manufactured by an Asian company and it boasts the largest market share domestically as one of the picosecond laser devices.
 Output is controlled according to the state and diagnosis of patients.
Output is controlled according to the state and diagnosis of patients. The applicator has patented ergonomic design, with the simple use of one-push types and streamlined design that helps smooth insertion. It is registered as a medical device class 2 in Korea and Japan, and various tests have been successfully completed such as vaginal irritation tests.
The applicator has patented ergonomic design, with the simple use of one-push types and streamlined design that helps smooth insertion. It is registered as a medical device class 2 in Korea and Japan, and various tests have been successfully completed such as vaginal irritation tests. LM/L400 features premium specifications with highest-level multi-function and remote-control option. The S300 is designed to be smaller in size, well reflecting the spirit of innovatively seeking “compactness,” and thus changing people’s preconceived ideas.
LM/L400 features premium specifications with highest-level multi-function and remote-control option. The S300 is designed to be smaller in size, well reflecting the spirit of innovatively seeking “compactness,” and thus changing people’s preconceived ideas.
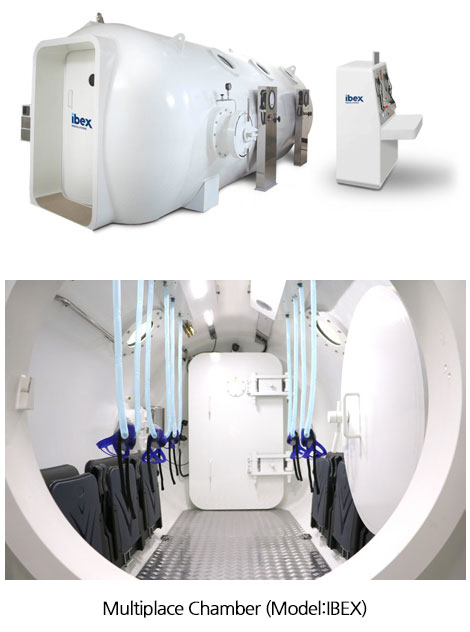
 maximize the effect. Each applicator is highly recognized for its convenience and safety for both user and patient.
maximize the effect. Each applicator is highly recognized for its convenience and safety for both user and patient.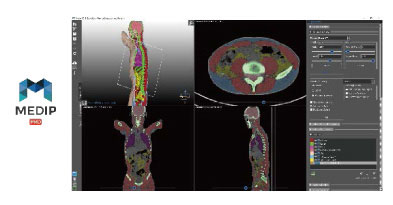
 MEDIP has drastically shortened 3D modeling time, and more technologies have been added and developed to enhance the functionality such as VR/AR, mobile communication service, and body composition analysis. ANATDEL, 3D-printed artificial organs, are very similar in size, texture, and elasticity to actual organs, so they are widely used for medical simulations of surgeons, education and practice of medical students, and testing medical devices. In particular, the demand for ANATDEL is expected to increase as a substitute for cadaver use.
MEDIP has drastically shortened 3D modeling time, and more technologies have been added and developed to enhance the functionality such as VR/AR, mobile communication service, and body composition analysis. ANATDEL, 3D-printed artificial organs, are very similar in size, texture, and elasticity to actual organs, so they are widely used for medical simulations of surgeons, education and practice of medical students, and testing medical devices. In particular, the demand for ANATDEL is expected to increase as a substitute for cadaver use.


